The outcome of this study was participant survival status, obtained from the national central registry system. Each participant was followed from their baseline registration date until either the recorded date of death or the study endpoint in December 2023, whichever came first.
This study analyzed four cardiometabolic markers: RBG, HbA1c, SBP, and DBP. RBG reflects short-term BG metabolism, while HbA1c provides a long-term measure, representing average BG levels over the past two to three months. RBG and HbA1c were measured from non-fasting blood samples collected at recruitment for all 500,000 participants, with a subset of 20,000 undergoing repeat assessments approximately five years later. RBG was analyzed using the hexokinase method, and HbA1c was measured via high-performance liquid chromatography. SBP and DBP were recorded using standardized devices, with the average calculated from two readings taken a few moments apart. WC was also measured by trained fieldworkers for each participant and categorized into four groups based on guideline-recommended thresholds: <88 cm, 88~102 cm, 102~116 cm, and >116 cm18.
A range of covariates was extracted from the UKB dataset, including: (1) sampling strata; (2) demographic variables such as age, race, education level, income level, and height; (3) lifestyle factors, including smoking history, drinking history, and physical activity status; and (4) comorbidity history, including hypertension, diabetes, and cardiovascular disease (CVD). Detailed definitions of these covariates are provided in Supplementary Table S1.
Statistical analysis
First, we constructed four separate fixed-effects Cox regression models to independently evaluate the associations between each of the four cardiometabolic markers (i.e., RBG, HbA1c, SBP, and DBP) and elderly mortality. The regression models can be parameterized as follows:
$$h(t)=_(t)\,exp(\beta x+{\boldsymbol{z}}+{\theta }_{i}),$$
where h(t) denotes the hazard function of mortality for each participant at the follow-up time t, x is the baseline level of cardiometabolic markers (i.e., RBG, HbA1c, SBP, or DBP, respectively), z indicates the covariates set, θi is the fixed effect of the sampling site, and β and γ are the corresponding coefficients. Specifically, BP values are strongly associated with participants’ history of hypertension, and BG values are closely linked to diabetes history, raising concerns about multicollinearity. To address this issue, and following the approach used in previous studies31,32, we included only hypertension history as a covariate in regression models assessing RBG and HbA1c, and only diabetes history in models assessing SBP and DBP. The missing values in the covariates were imputed using multivariate imputation by chained equations approach. HR = exp(β) was used to quantify the associations between cardiometabolic markers and mortality. Sex-specific effects of cardiometabolic markers were estimated using subgroup analysis.
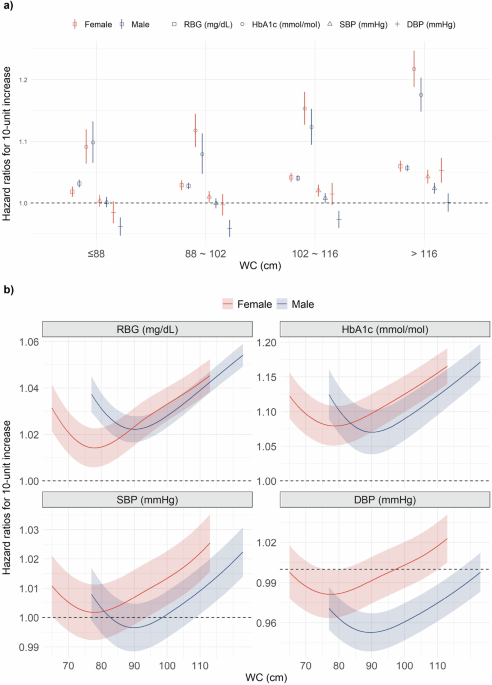
To assess the interaction effect between WC and cardiometabolic markers on elderly mortality, we first evaluated the heterogeneous linear associations between mortality and cardiometabolic markers by WC categories and sex. The HRs were estimated by including an interaction term between WC categories (c) and cardiometabolic markers (c:x) in the regression model as follows:
$$h(t)={h}_{0}(t)\,exp[\beta (c:x)+{\boldsymbol{\gamma }}{\boldsymbol{z}}+{\theta }_{i}].$$
Using a varying-coefficient model33, we further assessed the nonlinear modifying effect of WC by parameterizing the linear effect of cardiometabolic markers as a nonlinear function varying with different WC levels. This can be denoted as follows:
$$h(t)={h}_{0}(t)\,exp[f({x|}\mathrm{WC})+{\boldsymbol{\gamma }}{\boldsymbol{z}}+{\theta }_{i}],$$
where f(x | WC) denotes a nonlinear function of the cardiometabolic markers’ effect varying with different WC levels.
Second, previous studies have suggested nonlinear associations between mortality and BG or BP31,32. We further developed the sex-specific ERFs between mortality and cardiometabolic markers across different WC categories, to assess how the nonlinear associations between mortality and cardiometabolic markers varied by WC. The nonlinear ERFs were estimated using the following equation:
$$h(t)={h}_{0}(t)\exp [c:{\rm{ns}}(x,{\rm{df}}=5)+{\boldsymbol{\gamma }}{\boldsymbol{z}}+{\theta }_{i}],$$
where c:ns(x, df=5) denotes an interaction term between WC categories and natural spline function of cardiometabolic markers with five degrees of freedom.
Third, to directly assess the combined effects of WC and cardiometabolic markers on mortality, we developed a two-dimensional ERF. This approach models their joint impact by computing a tensor product interaction function, which offers greater flexibility than simple multiplicative interactions34. Unlike traditional models, tensor product functions allow for nonlinear relationships, making them particularly effective in capturing complex dependencies while remaining interpretable. The two-dimensional ERF enables direct estimation of mortality risk across varying combinations of WC and cardiometabolic markers. The model is specified as follows:
$$h(t)={h}_{0}(t)\exp [{te}({\rm{WC}},x)+{\boldsymbol{\gamma }}{\boldsymbol{z}}+{\theta }_{i}],$$
where te(WC, x) denotes a nonlinear tensor product interaction function between WC levels and cardiometabolic markers.
All statistical analyses were conducted using R (version 4.1.3; The R Foundation for Statistical Computing, Vienna, Austria). Statistical inference for the fixed-effects Cox regression model was performed using the R package survival, the natural spline function was developed using the R package splines, and the tensor product interaction function was developed using the R package mgcv.
link