Search results and study characteristics
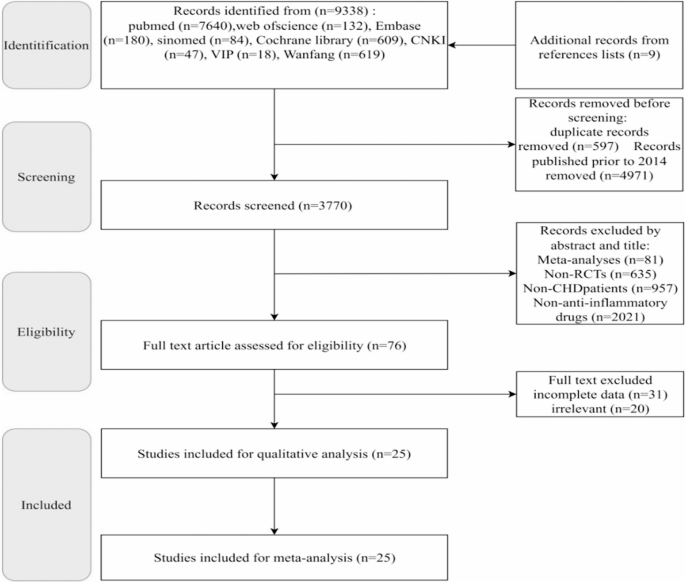
Initially, 9,329 relevant articles were retrieved from 8 databases, with an additional 9 articles identified from reference lists, totaling 9,338 articles. After excluding non-recent (older than 10 years) and duplicate publications, 3,770 articles remained for screening. Following title/abstract review and full-text assessment, a final total of 25 articles met the inclusion criteria [6,7,8,9, 14, 17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36]. The PRISMA flowchart is shown in Fig. 1.
The included studies comprised large-scale projects such as CANTOS and COLCOT, along with several smaller investigations. The new immunomodulatory drugs were utilized: anakinra (3 studies), colchicine (15 studies), tocilizumab (1 study), canakinumab (1 study), varespladib (1 study), darapladib (2 studies), methotrexate (1 study), and losmapimod (1 study). Baseline characteristics of the study populations are summarized in Table 1.
Risk of bias assessment
This systematic review included a total of 25 RCTs, rigorously assessed using the Cochrane Risk of Bias tool (RoB 2.0). The risk of bias assessment results are illustrated in Fig. 2. Details information is shown in supplement Fig. 2.
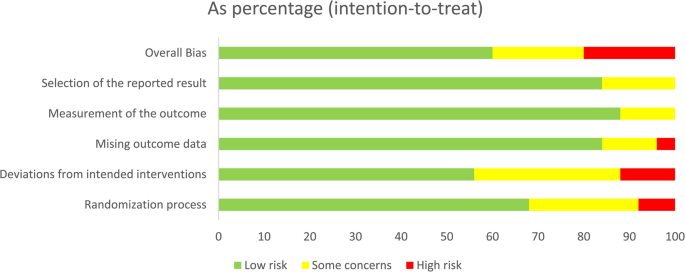
Risks of bias of the included studies. risk of bias. Green: low risk; yellow: some concern; red: high risk
Randomization process: most studies were judged as low risk, although a few raised some concerns, and two studies were judged as high risk [25, 35]. Deviations from intended interventions: 8 studies were rated as having some concerns [17]– [18, 25,26,27, 29, 35]– [14], and 3 studies were rated as high risk [19, 22, 36], primarily due to the open-label design without placebo control. Missing outcome data: most studies were rated as low risk because all randomized participants completed follow-up. One study was considered high risk due to a high rate of loss to follow-up [34]. Measurement of the outcome: The majority of studies were judged as low risk, particularly those employing blinded outcome assessment; only three studies were rated as having some concerns [19, 25, 36]. Selection of the reported result: Four studies raised some concerns, mainly due to unavailable preregistered protocols or potential selective reporting [23,24,25, 29].
Primary outcome
The primary outcome of this study was the incidence of MACE. Subgroup analyses were conducted based on the type of new immunomodulatory drugs, follow-up duration, and disease classification, with results detailed in Fig. 3.
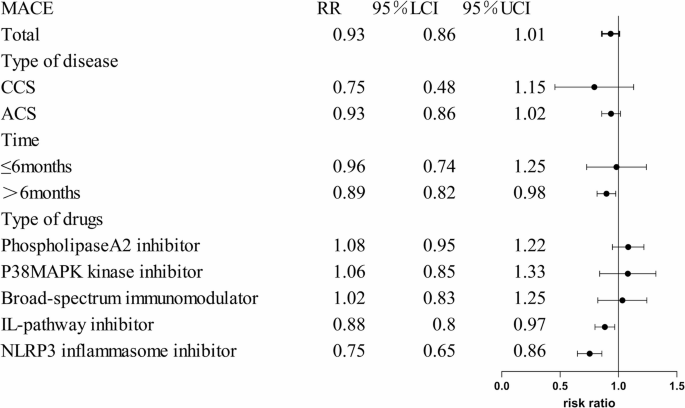
Subgroup analysis of MACE
Overall summary
Among the 25 included studies, 15 reported the incidence of major adverse cardiovascular events (MACE) [6,7,8,9, 14, 17, 20, 26,27,28,29,30, 32,33,34]. Adding new immunomodulatory drugs to standard therapy during treatment did not show significant improvement in MACE incidence compared to standard therapy alone. (RR = 0.92,95%CI: [0.84,1.01], P=0.09, I²=60%, 17 trials, 65,420 participants).
Subgroup analysis based on the types of new immunomodulatory drugs
Based on different immunomodulatory drugs, these studies were divided into the IL-pathway inhibitors, NLRP3 inflammasome inhibitors, Lipoprotein-associated phospholipase A2 (Lp-PLA2) Inhibitors, p38 Mitogen-Activated Protein Kinase (MAPK) Inhibitors, broad-Spectrum immunomodulators. Compared with the control group, the incidence of MACE was significantly reduced in the NLRP3 inflammasome inhibitors group (RR = 0.75, 95% CI: [0.65,0.86], P < 0.0001), and the IL-pathway inhibitors group (RR = 0.86, 95% CI: [0.75,0.97], P = 0.02). However, in the Broad-spectrum immunomodulator group, Lp-PLA2 inhibitor group, and p38 MAPK kinase inhibitor group, no significant improvement in MACE incidence was observed compared with the control group (P > 0.05). The forest plot are shown in Supplement Fig. 2.1.
Subgroup analysis based on the follow-up time
Based on the follow-up periods, these studies were divided into a group with follow-up ≤ 6 months and a group with follow-up > 6 months. Compared with the control group, when the follow-up period was ≤ 6 months, the improvement in MACE incidence in the intervention group was not significant (P > 0.05), whereas when the follow-up period exceeded 6 months, the intervention group showed a significant reduction in MACE incidence (RR = 0.88, 95% CI: [0.79,0.97], P = 0.01). The forest plot are shown in Supplement Fig. 2.2.
Subgroup analysis based on the disease classification
Based on the different disease types of the included population, these studies were divided into an ACS group and a CCS group. Compared with the control group, the MACE incidence rate in the intervention group with CCS showed no significant improvement (P > 0.05), and similarly, the incidence of MACE in the intervention group with ACS also showed no significant improvement (P > 0.05). The forest plot are shown in Supplement Fig. 2.3.
Secondary outcome
The secondary outcomes of this study included the incidence of, incidence of cardiac arrest (CA), incidence of gastrointestinal adverse reaction, incidence of death from any cause, incidence of revascularization, incidence of infection, hs-CRP, left ventricular ejection fraction (LVEF), IL-6, infarct size, and neutrophil count. The specific details are provided in the Table 2.
Incidence of CA
Three studies [7, 26, 32] reported the cardiac arrest. The comparison of the cardiac arrest indicator between the intervention group and placebo group showed no statistically significant difference (RR = 0.87, 95% CI: [0.38, 2.01], P = 0.75). These results show that new immunomodulatory drugs could not reduce the incidence of CA compared with different placebo groups. The forest plot are shown in Supplement Fig. 3.
Incidence of angina
Ten studies [6, 7, 14, 17, 20, 26, 28, 32, 34, 36] (13 trials) reported angina incidence. The results showed that compared with the placebo group, the incidence of angina in the intervention group were markedly improved, with a statistically significant difference (RR = 0.73, 95% CI: [0.58,0.92], P = 0.007). These results show that new immunomodulatory drugs could reduce the incidence of angina compared with different placebo groups. The forest plot are shown in Supplement Fig. 4.
Incidence of all-cause mortality
Thirteen studies [6,7,8,9, 14, 17, 18, 20, 27,28,29, 33, 34] (16 trials) reported the incidence of death from any cause. The results showed no statistically significant difference in the death from any cause outcome between the intervention group and placebo group (RR = 0.98, 95% CI: [0.92, 1.04], P = 0.50). These results show that new immunomodulatory drugs could not reduce the incidence of all-cause mortality compared with different placebo groups. The forest plot are shown in Supplement Fig. 5.
Incidence of revascularization
Nine studies [6, 8, 9, 14, 17, 29, 30, 33, 34] (11 trials) reported the revascularization indicators. The outcomes demonstrated that the incidence of revascularization in the intervention group reduced with a statistically significant difference (RR = 0.86, 95% CI: [0.74,0.99], P = 0.04) compared with the different control group. These results show that new immunomodulatory drugs could reduce the incidence of revascularization compared with different placebo groups. The forest plot are shown in Supplement Fig. 6.
Incidence of gastrointestinal adverse effect
Four studies [7, 32, 33, 36] (5 trials) reported the incidence of gastrointestinal adverse effect. The incidence of gastrointestinal adverse showed no statistically significant difference between the intervention group and placebo group (RR = 1.22, 95% CI: [0.85, 1.74], P = 0.27). These results show that new immunomodulatory drugs could not reduce the incidence of revascularization compared with different placebo groups. The forest plot are shown in Supplement Fig. 7.
Incidence of infection
Seven studies [6,7,8,9, 18, 27, 29] encompassing 10 trials reported the incidence of infection. There was no statistically significant difference between the intervention and placebo groups (RR = 1.06, 95% CI: 0.92–1.22, P = 0.45). These findings indicate that the new immunomodulatory drugs did not reduce the risk of infection compared with various placebo controls. The corresponding forest plot is presented in Supplementary Fig. 12.
Inflammatory markers
Nine [19, 21,22,23,24, 26, 27, 35, 36] studies (10 trials) reported the hs-CRP. The results showed that there was no significant difference of hs-CRP between placebo group and intervention group (MD=−1.05, 95% CI: [−2.10,0.00], P = 0.05). The forest plot are shown in Supplementary Fig. 8. A total of 3 studies [25, 27, 36] (5 trials) reported IL-6. The results showed that IL-6 was markedly reduced in intervention group when compared with the different placebo groups, (MD=−4.11, 95% CI: [−7.13, −1.09, P = 0.008). The forest plot are shown in Supplementary Fig. 9. A total of 2 studies [22, 36] (3 trials) reported neutrophil count. The results showed that the neutrophil count in the intervention group were markedly reduced compared with the different placebo groups, (MD=−0.74, 95% CI: [−1.19, −0.29], P = 0.001). The forest plot are shown in Supplementary Fig. 10. These results show that compared with different placebo groups, the new immunomodulatory drugs could not reduce level of the hs-CRP, but could reduce the level of IL-6 and neutrophil count.
LVEF
Two studies [26, 36] (3 trials) reported data on left ventricular ejection fraction. The comparison of LVEF between the intervention group and different placebo groups showed no statistically significant difference (MD = 1.41, 95% CI: [0.08,2.75], P = 0.04). These results show that new immunomodulatory drugs were associated with an increase in LVEF compared with various placebo groups. The forest plot are shown in Supplementary Fig. 11.
Publication bias, meta-regression, and sensitivity analysis
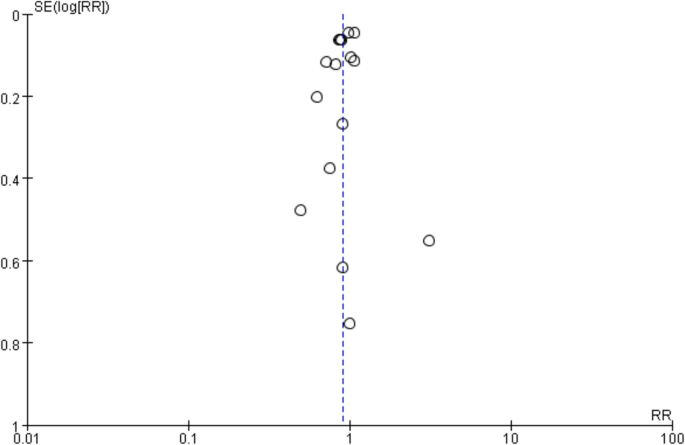
The funnel plots are shown in Fig. 4, the results showed that the funnel plot was symmetrical, suggesting the absence of publication bias. In addition, both Egger’s test (P = 1.00) and Begg’s test (P = 0.84) indicated P > 0.05, further supporting the conclusion that publication bias is unlikely.
Meta-regression indicated that type of drugs is a significant source of heterogeneity, P = 0.002. The country class and year are not a significant source of heterogeneity, P>0.05.
In the sensitivity analysis, the point estimate for MACE remained stable (RR range: 0.91–0.95) regardless of which individual study was omitted, indicating the overall result was robust. Details information is shown in Table 3. The exclusion of certain large trials, such as Nicholls 2014 or Nidorf 2020, led to minor fluctuations in the pooled risk ratio and its confidence intervals, but these changes were not substantial enough to alter the primary conclusion of the analysis. For instance, upon excluding Nicholls 2014, the result became statistically significant (RR = 0.90, 95% CI: 0.82, 0.98, P = 0.02), while excluding Nidorf 2020 yielded an RR of 0.94 (95% CI: 0.86,1.03, P = 0.20). The fact that no single study dramatically changed the effect estimate suggests that the observed heterogeneity (I² = 60%) is not driven solely by any one trial but is more likely attributable to broader differences across studies, such as variations in drug mechanisms of action, as identified in our subgroup analysis and meta-regression.
Quality of the evidence
This study conducted GRADE evidence ratings for the primary outcome and secondary outcomes. The evidence quality for the incidence of MACEs was accessed as moderate. This indicates that the current evidence is highly credible regarding the efficacy and safety of new immunomodulatory drugs therapy in patients with CHD. Detailed results are presented in Table 4.
link

