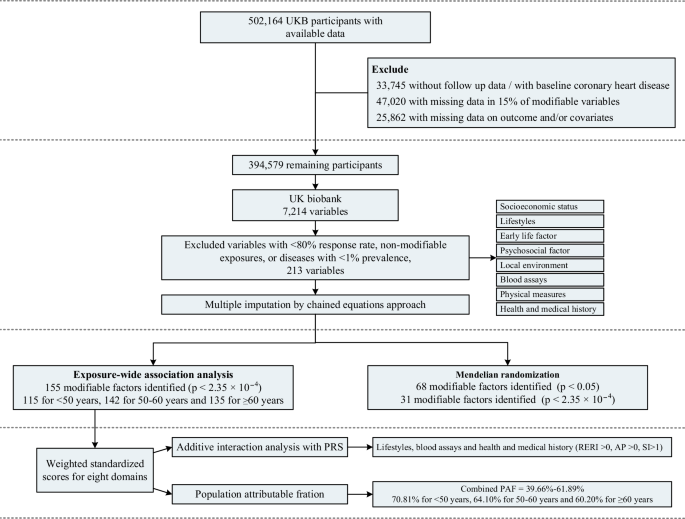
This study included 394,579 participants (56.1% females; Supplementary Data 4), with a mean (SD) age of 56.1 (8.1) years at recruitment. After 13.9 (2.3) years of follow-up, 27,592 participants were diagnosed with CHD, with a mean (SD) age of diagnosis at 68.4 (7.6) years. This study identified 155 out of 213 modifiable factors associated with CHD (Fig. 2 and Supplementary Data 5). These factors were categorized into eight domains: socioeconomic status (SES, 7 factors), lifestyles (32 factors), early life factors (2 factors), psychosocial factors (7 factors), local environment (8 factors), blood assays (37 factors), physical measures (10 factors), and health and medical history (52 factors). Among these, 33 factors were identified as protective factors, and 122 were identified as risk factors. According to the variable importance rankings from the random forest model, the top 10 predictors were: diabetes mellitus [hazard ratio (HR) = 2.04, 95% confidence interval (CI): 1.96, 2.13, p < 2.74 × 10−265], Cystatin C (HR = 1.18, 95% CI: 1.17, 1.19, p < 2.35 × 10−04), long-standing illness, disability or infirmity (HR = 1.73, 95% CI: 1.69, 1.78, p < 2.35 × 10−04), Overall health rating (HR = 1.42, 95% CI: 1.36, 1.47, p = 2.87 × 10−67 for “good”, HR = 2.37, 95% CI: 2.27, 2.48, p < 2.35 × 10−04 for “fair,” HR = 4.01, 95% CI: 3.79, 4.25, p < 2.35 × 10−04 for “poor”; all compared with excellent), systolic blood pressure (HR = 1.20, 95% CI: 1.18, 1.22, p = 8.90 × 10−118), testosterone (HR = 0.84, 95% CI: 0.81, 0.88, p = 8.43 × 10−16), glycated hemoglobin (HR = 1.12, 95% CI :1.12, 1.13, p < 2.35 × 10−04), leg fat percentage (HR = 2.33, 95% CI: 2.23, 2.44, p = 4.23 × 10−300), attendance/disability/mobility allowance (HR = 2.11, 95% CI: 2.03, 2.20, p = 1.15 × 10−275), and HDL cholesterol (HR = 0.72, 95% CI: 0.70, 0.73, p = 2.30 × 10−252).
A The x-axis represents the hazard ratio value, and the y-axis represents statistical significance (that is, the 30th root of the p value). The dashed line represents the threshold after multiple testing correction (Bonferroni correction, p value < 2.35 × 10−04). The point size represents minimal depth, which was calculated by simultaneously including 213 modifiable factors and covariates in a random forest model with Cox proportional hazards regression to rank the importance of the variables; smaller minimal depth indicates greater variable importance. A set of the highest-risk factors was annotated. B The points represent hazard ratios, and the horizontal lines represent the corresponding 95% confidence interval. The rank indicates the importance of the variable in order of minimal depth. Hazard ratios were calculated using Cox proportional hazards regression analysis, adjusted for baseline age, sex, ethnic background, family history of cardiovascular disease, and assessment center (N = 394,579). Two-sided Z-tests were used to assess statistical significance. ATHI average total household income before tax, HFD/SP hands-free device/speakerphone use with mobile phone, VIT B9 Vitamin B9, HbA1c hemoglobin A1c, HDL high-density lipoprotein, LDL low-density lipoprotein, IGF-1 Insulin-like growth factor 1, MET metabolic equivalent of task, SHBG sex hormone-binding globulin. Source data are provided as a Source data file (Supplementary Data 5).
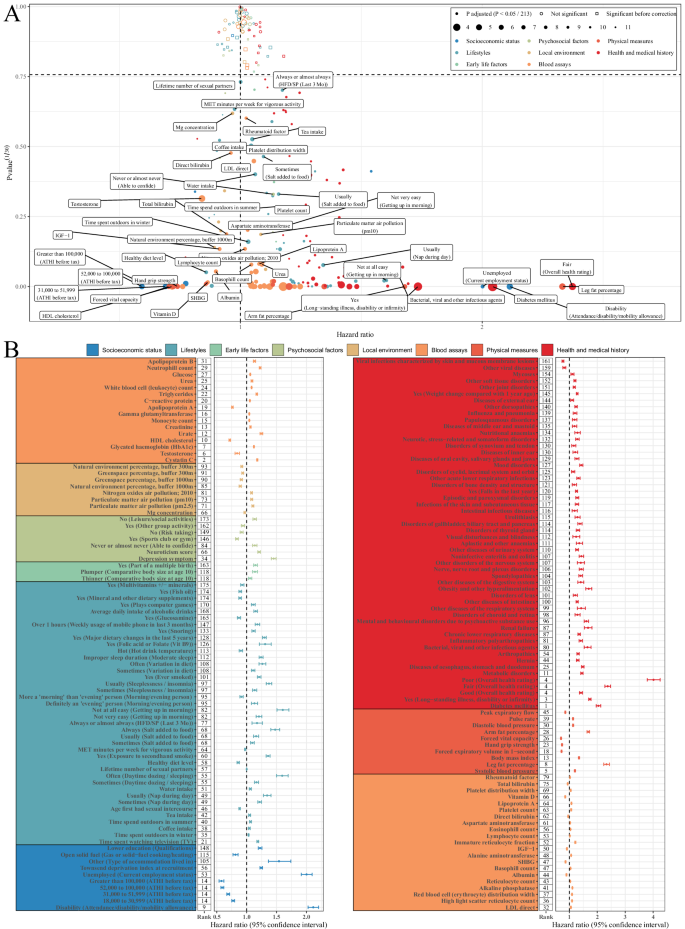
Further analyses stratified by age, sex, genetic risk for CHD, and follow-up duration revealed that >100 factors remained stably associated with CHD in most subgroups, except for the group with >10 years of follow-up, although the specific associations varied across subgroups (Fig. 3, Supplementary Data 6 and Supplementary Figs. 1–9). The number of modifiable factors associated with CHD increased with age, yet the effects of most factors decreased. Moreover, older populations showed greater sensitivity to local environmental factors. Variable importance rankings from the random forest model showed broadly consistent results across subgroups, with factors in health and medical history, blood assays, and physical measures consistently ranking high in importance, while the importance of factors in other domains diminished with age.
Models were estimated using Cox proportional hazards regression, adjusted for baseline age, sex, ethnic background, family history of cardiovascular disease, and assessment center (N = 394,579). Two-sided Z-tests were used to assess statistical significance. The color of cells indicates the effect sizes between each risk factor and incident coronary heart disease. Asterisks in cells represent significant associations after correction for multiple testing (Bonferroni-corrected, p < 2.35 × 10−04). ATHI average total household income before tax, HFD/SP hands-free device/speakerphone use with mobile phone, VIT B9 Vitamin B9, HbA1c hemoglobin A1c, HDL high-density lipoprotein, LDL low-density lipoprotein, IGF-1 Insulin-like growth factor 1, MET metabolic equivalent of task, SHBG sex hormone-binding globulin. Source data are provided as a Source data file (Supplementary Data 6).
Inverse variance-weighted MR was employed as the primary analysis after removing outliers (Fig. 4, Supplementary Data 7, and Supplementary Figs. 11–14), given the observed evidence of heterogeneity (p < 0.05 for the Q test) and horizontal pleiotropy (p < 0.05 for the Egger intercept). By the threshold of p < 0.05, 68 factors showed associations with CHD. At a more stringent threshold of p < 2.35 × 10−04, 31 factors demonstrated causal relationships with CHD, including 22 factors exhibiting positive associations and 9 with negative associations. These factors spanned six domains: blood assays (13 factors, e.g., triglycerides), early life factors (2 factors, e.g., comparative body size at age 10), health and medical history (5 factors, e.g., self-reported hypertension), lifestyle behaviors (3 factors, e.g., alcohol intake frequency), physical measures (6 factors, e.g., waist circumference), and SES (2 factors, e.g., educational attainment). Results from alternative methodologies are presented in Supplementary Data 7.
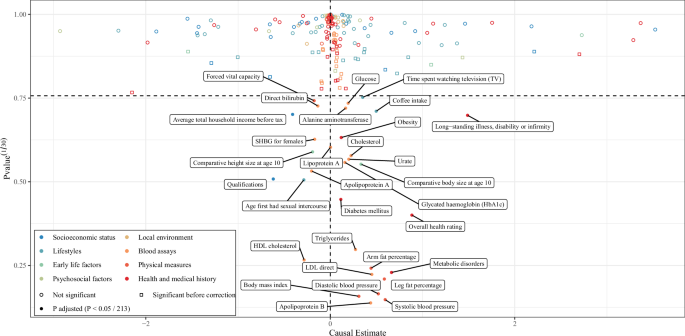
Causal estimates were generated using the inverse variance-weighted method after removing outliers. The dashed line represents the threshold after multiple testing correction (Bonferroni correction, p value < 2.35 × 10−04). Two-sided statistical tests were performed. Dots represent ln (odds ratios). A set of the highest-risk factors was annotated. Source data are provided as a Source data file (Supplementary Data 7).
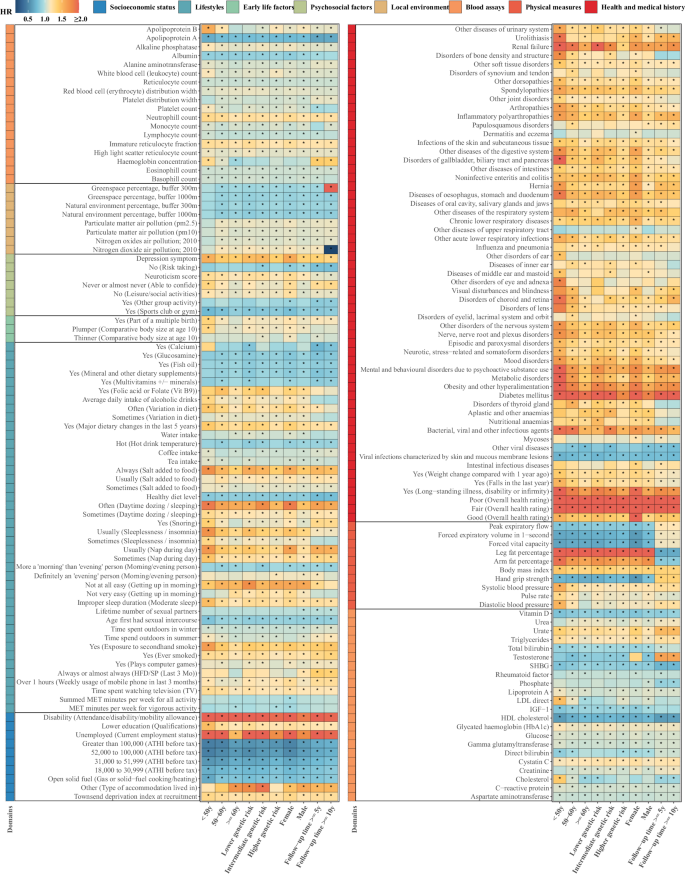
Compared to favorable profiles (Table 1), both unfavorable early life and psychosocial factors, as well as intermediate or unfavorable profiles across SES, lifestyle, local environment, blood assays, physical measurements, and health and medical history, were significantly associated with higher CHD risk. A significant increasing trend in CHD risk was observed across all eight domains (p for trend <0.001). Stratified analysis by age (<50 years, 50–60 years, and ≥60 years) revealed that the adverse effects of intermediate and unfavorable profiles across SES, lifestyles, early life factors, psychosocial factors, blood assays, as well as health and medical history diminished with increasing age (Fig. 5 and Supplementary Data 8). Additionally, a multiplicative interaction between age and the weighted score for health and medical history demonstrated that the effect of having a moderate or unfavorable health and medical history score on CHD risk gradually diminished with increasing age (p for interaction <0.05, Supplementary Data 8).
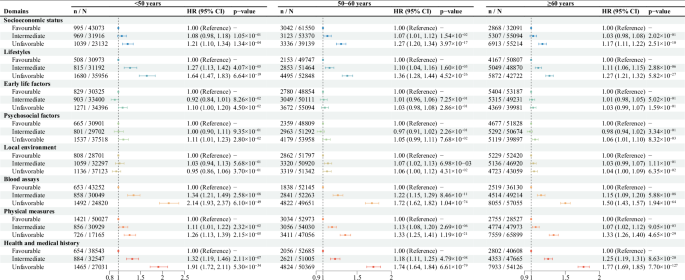
The favorable profile was set as a reference in each domain. The associations were estimated using a Cox proportional hazards regression model that included all eight domains, mutually adjusted, and with adjustments for baseline age, sex, ethnic background, family history of cardiovascular disease, and assessment center. The points represent hazard ratios (HR), and the horizontal lines represent the corresponding 95% confidence interval (CI). Two-sided statistical tests were used. N number of individuals at risk, n number of CHD cases. Source data are provided as a Source data file (Supplementary Data 8).
Meanwhile, participants were divided into nine groups based on tertiles of PRS and weighted scores for each domain, respectively. As shown in Fig. 6, Supplementary Data 9 and Supplementary Figs. 15–17, individuals with the highest genetic risk scores and unfavorable weighted scores had substantially increased CHD risks compared with those with the lowest genetic risk scores and favorable weighted scores: 134% higher for SES, 157% for lifestyles, 94% for early life factors, 97% for psychosocial factors, 96% for local environment, 192% for blood assays, 162% for physical measures, and 240% for health and medical history. Statistically significant interactions were observed between weighted scores for lifestyles, blood assays, and health and medical history with PRS, which diminished with age (Supplementary Data 10).
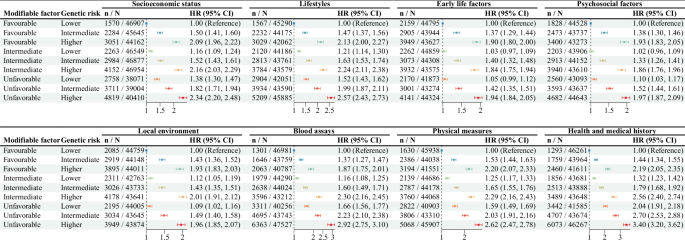
Participants were categorized into nine groups based on the tertiles of the PRS, combined with domain-weighted scores. Hazard ratios were generated using a Cox model that included all eight domains, mutually adjusted, and with adjustments for baseline age, sex, ethnic background, family history of cardiovascular disease, and assessment center. The points represent hazard ratios (HR), and the horizontal lines represent the corresponding 95% confidence interval (CI). Two-sided statistical tests were used. The additive interaction results of the PRS and the weighted scores for each domain are available in Supplementary Data 9 and 10. N number of individuals at risk, n number of CHD cases. Source data are provided as a Source data file.
When shifting all unfavorable profiles to intermediate and favorable ones (Model 1), PAF estimation suggested that 39.66% of CHD cases could be prevented (Table 2), increasing to 61.89% when shifting all factors to the favorable tertile. In the conservative assessment (Model 1), the estimated maximum preventive effect was attributed to health and medical history, resulting in an 11.37% reduction in CHD incidence. The contributions of remaining domains were described as follows: blood assays (9.54%), lifestyles (5.97%), physical measures (5.70%), SES (3.70%), psychosocial factors (2.06%), early life factors (0.99%), and local environment (0.33%). In the context of a feasible implementation of comprehensive elimination of these factors, health and medical history would still account for the highest proportion of prevented CHD cases (15.75%), followed by blood assays (13.87%) and lifestyles (10.01%). Notably, implementing preventive measures in younger populations (under the age of 50), including blood indicator monitoring, healthy lifestyles, and disease prevention, could yield substantial benefits, potentially preventing 49.34–70.81% of CHD cases (Supplementary Data 11–13). Two sensitivity analyses were conducted, calculating PAFs by including only factors significantly associated with CHD (p < 0.05), and further adjusting for PRS when estimating PAFs. Both approaches yielded results consistent with the main findings (Supplementary Data 14 and 15). Additionally, considering the potential temporal ordering among different domains, PAFs were also calculated using models without mutual adjustment between domains, suggesting the possibility of potential prevention of an even larger proportion (59.77–77.65%) of CHD cases (Supplementary Data 16).
link