We conducted a DCE in accordance with the relevant guidelines and regulations provided by the International Society of Pharmacoeconomics and Outcomes Research (ISPOR) good research practices that aimed to standardize the methods used for conjoint analysis in healthcare research12. Each respondent was asked to answer ten tasks; each task consisted of a multiple-choice question with three alternatives from which he had to select the one that perceived maximizing the utility from his perspective. Each alternative had a different profile based on the levels of the attributes. The alternative’s attributes consisted of a set of drug classes (a whole regimen), and their levels were whether the drug class was a brand or generic.
Study population
The study included a sample of adult nationally insured CHD patients in Alexandria, Egypt, who regularly come to the health insurance clinics in Alexandria to get their medicines. Alexandria has one of the highest HIO coverage in Egypt at 77.2% which indicates the importance of studies at HIO institutions as they represent an important sector of Alexandrian population13. The study included the following drugs given for the secondary prevention of CHD: hydroxyglutaryl-CoA reductase inhibitors (statins), beta-blockers, RAAS blockers, and aspirin. The HIO offers all generics free of charge and the brands for a charge calculated from the difference between the procurement costs of the brands and their generics. Patients with psychological problems or mental disabilities were excluded.
The following methodological sections address the recommended ISPOR checklist for conjoint analysis in health care including the attributes and levels, construction of tasks, experimental design, preference elicitation, instrument design, data collection, and statistical analyses. In addition, a section describing how drivers for brand inclination was assessed is included before the statistical analyses.
Identification and selection of attributes and their levels
We identified six classes with proven benefits in reducing cardiovascular morbidity and mortality; the list included statins, beta-blockers, RAAS blockers, antiplatelets (PGY12 inhibitors and aspirin), and aldosterone antagonists14,15,16. After reviewing the prescription norms in HIO, we found that aspirin-PGY12 inhibitor dual antiplatelet therapy was prescribed for only one year post-acute coronary syndrome (post-ACS), and patients were de-escalated to aspirin alone. Additionally, we found that a minority were prescribed an aldosterone antagonist as indicated in CHD only for patients with LVEF ≤ 40% post-ACS who received a beta-blocker and ACE inhibitor. Therefore, we only included the four agents prescribed to most CHD patients: statins, beta-blockers, RAAS blockers, and aspirin. Each selected attribute (drug class) was set to have one of two levels (brand or generic).
A price for each alternative was calculated based on the classes whose levels were set to brands. Each class’s brand had a different price representing the average out-of-pocket amount: the procurement prices of the class’s brands minus the class’s generic procurement price available in the HIO. Since we had four classes, each with two levels, this resulted in 16 (2^4) different alternatives (profiles) with 16 different prices ranging from EGP 0 (when all classes’ levels were set to “generic”) to EGP 285 (when all classes’ levels were set to “brand”). The sixteen possible prices set for each profile according to the combination of class levels were EGP 0, EGP 22, EGP 41, EGP 63, EGP 68, EGP 90, EGP 109, EGP 131, EGP 154, EGP 176, EGP 195, EGP 217, EGP 222, EGP 263, EGP 244, and EGP 285.
Construction of tasks
Each task consisted of three alternatives: two generated by the Conjointly software, and the third one was the status quo of the patient. The status quo is a term used to describe the respondent’s current situation; in our study, the status quo represents the patient’s combination set of the free-of-charge generics and the out-of-pocket brands he purchases.
Experimental design
We used a D-efficient main effects design generated by Conjoint.ly software. This fractional factorial design has optimized orthogonality, level balance, and minimal overlap. The design included 16 blocks randomly assigned to the respondents, and each block consisted of 10 choice tasks without the price attribute. The total number of alternatives in the 16 blocks was 320 (20 alternatives per block). We then calculated the price of each of the 320 alternatives according to the classes of medicines whose levels were set to “brand”; this represented a realistic price and aimed to prevent the generation of choices with unbalanced utilities. Finally, we added the status quo for each respondent to each choice task during the interview.
Preference elicitation
To increase the statistical efficiency of our design, we presented two versions for each choice task: an initial version in which the prices were set as mentioned in the construction of tasks section and an adapted version where the prices of the two software-generated alternatives were decreased or increased depending on the alternative chosen in the initial version. If the respondent chose the cheapest alternative in the initial version, we asked the respondent to decrease the prices of the two unchosen alternatives down to a price that would change his initial choice to one of the originally unchosen alternatives. In contrast, if the respondent chose the most expensive alternative, we asked him to state the maximum price of this alternative he would pay. Figure 1 illustrates an example of an initial choice task before price adaptation from which the respondent chose the alternative that maximized the utility from his perspective; here, the respondent chose the cheapest alternative (with all classes’ levels set to “generic”, marked with ↑) and the adapted version where the prices of the two originally unchosen alternatives were reduced until he changed his choice to one of them.
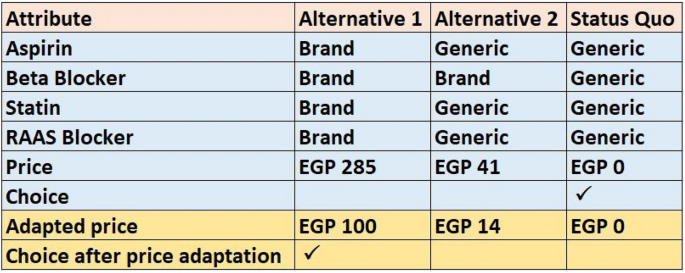
Example of an answered choice task before and after price adaptation.
Instrument design and data collection
The choice set cards were constructed using visual aids. The interviewer manually distributed the actual packs of the medicines into two groups representing the two alternatives on each card. Each group included only the classes whose levels were set to “brand” in the alternative. The interviewer asked the patient whether he preferred the brands of group 1 priced at price x or to get the brands of group 2 at price y more than his status quo. The interviewer recorded the responses to each task on the data collection sheet corresponding to the block without exhausting the patient. We repeated one of the tasks to check the consistency and quality of the responses; only results with consistent choices were included in the analysis.
Drivers for brand inclinations
We surveyed the patients for their drivers of brand inclination for each of the four CHD medications; we first identified the respondents who were not satisfied with the generic and then asked them to state the cause of dissatisfaction that drove them to be inclined to the brand. The drivers of brand inclinations were grouped into two general drivers: internal and external. The internal drivers included the insufficient efficacy of the generic, the side effects of the generic, or the general belief in the better quality of the brands. The external drivers included the advice of an HIO physician, other physicians, or a friend or relative.
Sample size calculation
The sample size calculation was done using Conjointly software17 by simply entering the attributes (five attributes), levels (brand or generic), number of questions per participant (ten choice sets), and the number of alternatives in each question (two alternatives per choice set). This resulted in a minimum required sample size of 100 participants for the main analysis. We targeted 208 respondents (13 respondents for each block of the 16 blocks) to allow for subgroup analyses.
Statistical analysis
Categorical variables were described using frequencies and percentages. Continuous variables were described using the mean and standard deviation. Independent t tests and chi-squared tests were performed to compare the sociodemographic characteristics of the satisfied and dissatisfied respondents.
Conditional logistic regression was used to estimate patients’ preference weights for the classes’ brands, the attribute part-worth utility of the whole class, and the willingness to pay (WTP) for the brand of each class of medicines. CHD classes’ levels had only two levels: brand and generic, where the generic was the reference. The preference weights β of the classes represent the change in the utility from generic to brand. For the price attribute, β represents the change in utility for every one-unit increase in the price of the regimen. Positive preference weights mean the brand is more preferred than the generic, and negative weights mean the opposite. The adjusted odds ratio (AOR) was calculated as the exponent of β and represented the odds of achieving higher utility.
Attribute part-worth (relative importance score) represents the average relative effect of each attribute on the change in the regimen’s utility and it is defined as the absolute difference between the highest and lowest coefficients in the range of preference weights within that attribute, divided by the total of the ranges of preference weights within all other attributes (including the price). In comparison to the other attributes in the model, it indicates the degree to which that attribute affects overall utility18. The WTP values represent the average amount of money in EGP the respondents are willing to pay to purchase the brand of each of the four CHD drug classes included in the DCE. Negative values mean the brand is less preferred than the generic for the corresponding class. To calculate the attribute partworths, we multiplied the preference weight of the price by 285 to account for the highest price presented to the respondents because the price is a continuous attribute. This adjustment estimates the change in utility between the best and worst prices.
We conducted four subgroup analyses based on sex, age, education, and income. For age, we split the sample of the dissatisfied respondents into two groups: above and below median age. We created two education groups: one with at least intermediate education and one below intermediate education. Finally, we split the sample into two income groups: one in debt and one at least meeting the routine expenses (not in debt).
R Software version 4.1.1.19 was used for data analysis and modeling. The mlogit package version 1.1-120 was used to conduct conditional logistic regression.
Ethical considerations
The study and all experimental protocols were approved by the HIO administration and the research and ethics committee of the central directorate for research and health development in the Egyptian Ministry of Health and Population. All methods were performed in accordance with relevant guidelines/regulations. An informed consent was obtained from all subjects and/or their legal guardian(s), after providing them with information about the study purpose.
link

